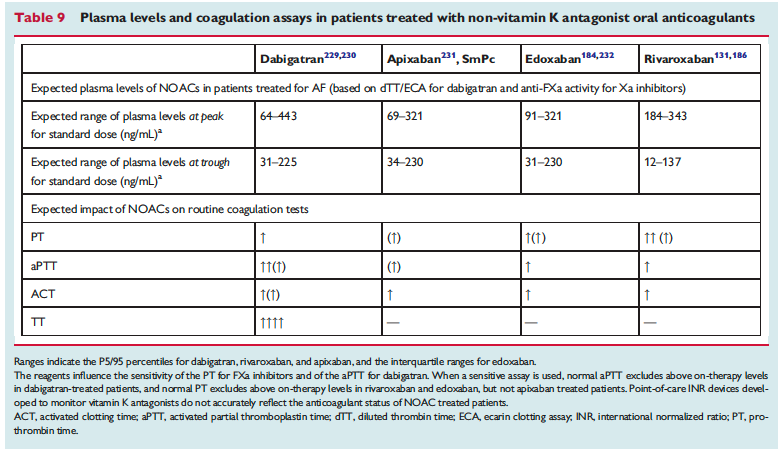
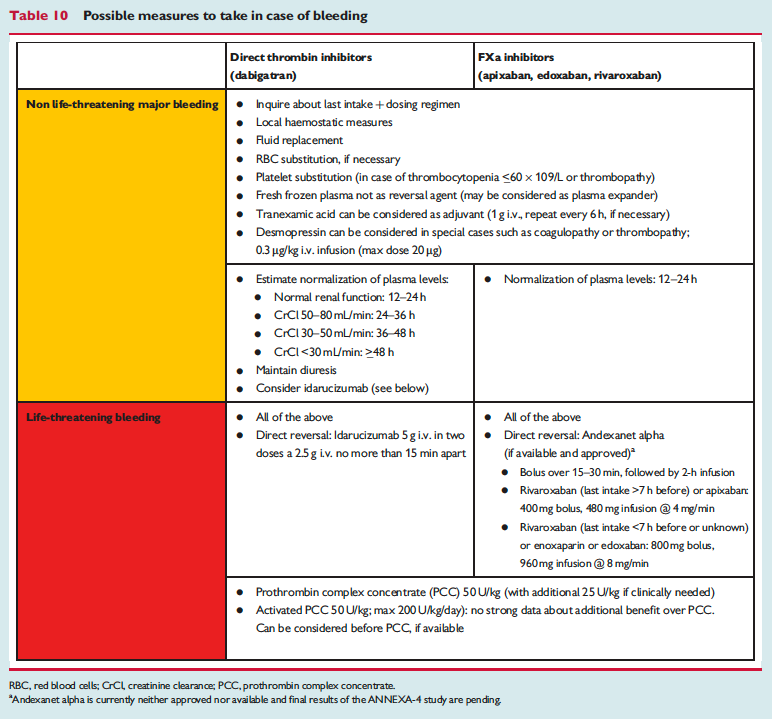
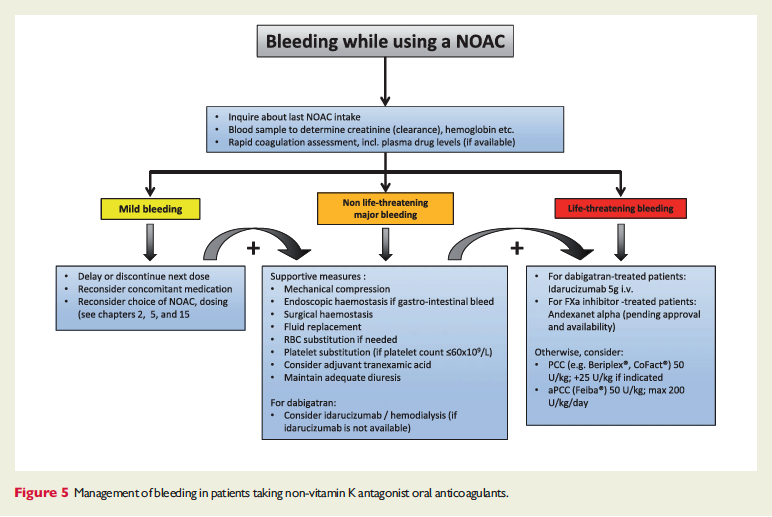
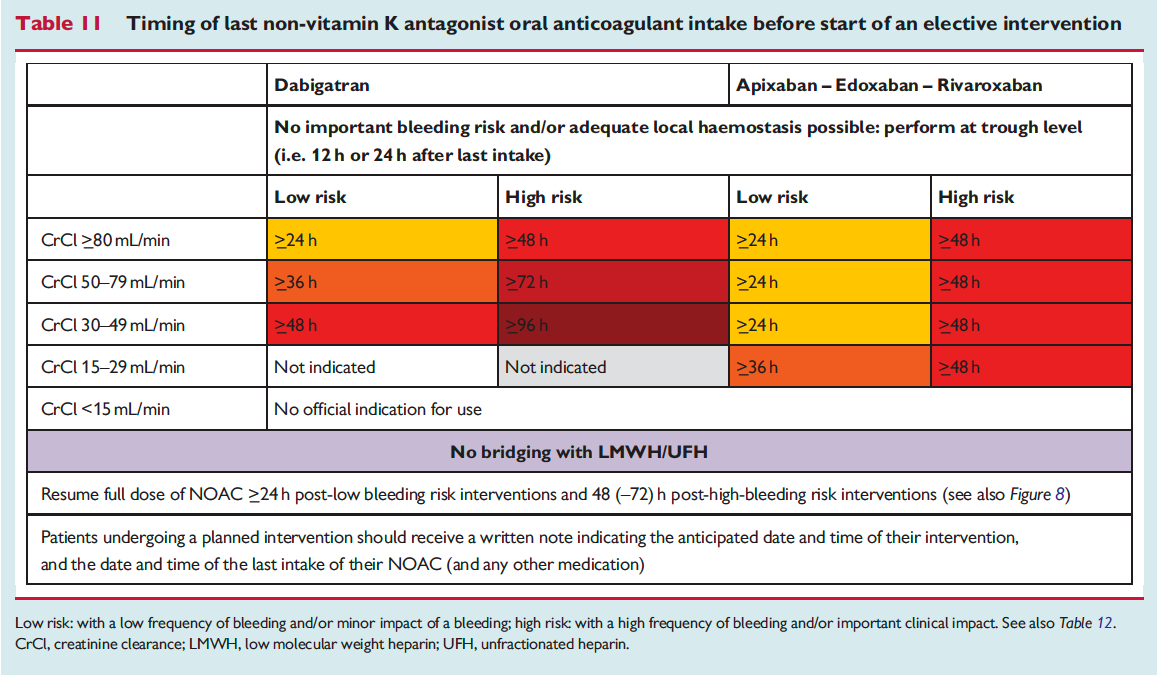
The European Heart Rhythm Association has published an updated set of NOAC guidelines. When these guidelines were first published 2 years ago, they were really the most in-depth set of recommendations out there and went places that the FDA was too afraid to go (i.e actual advice!). Common questions are addressed – NOAC selection, drug-drug interactions, levels, renal function, time off of NOAC for procedure, reversal (adnexanet even gets an appearance now along with Idarucizumab!). You may not agree with all of these suggestions but they are at least a great starting place.
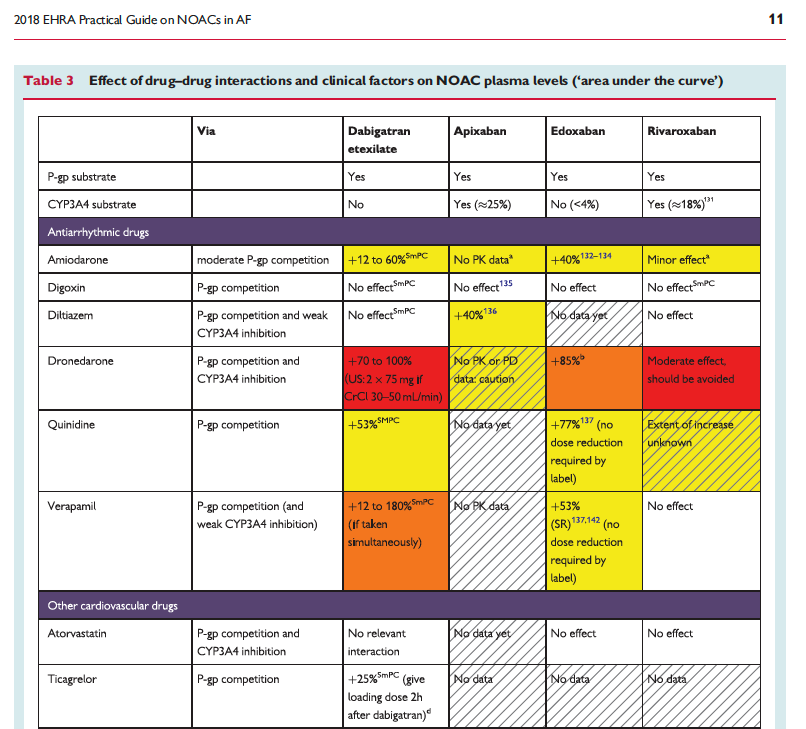
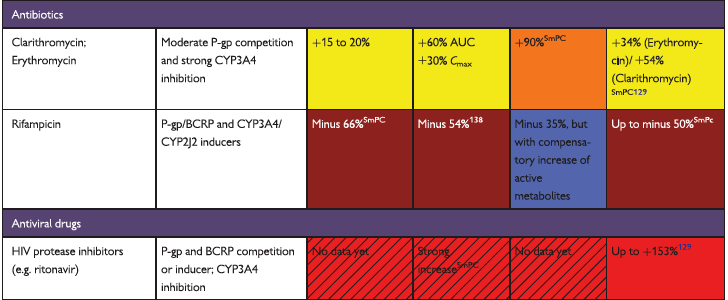
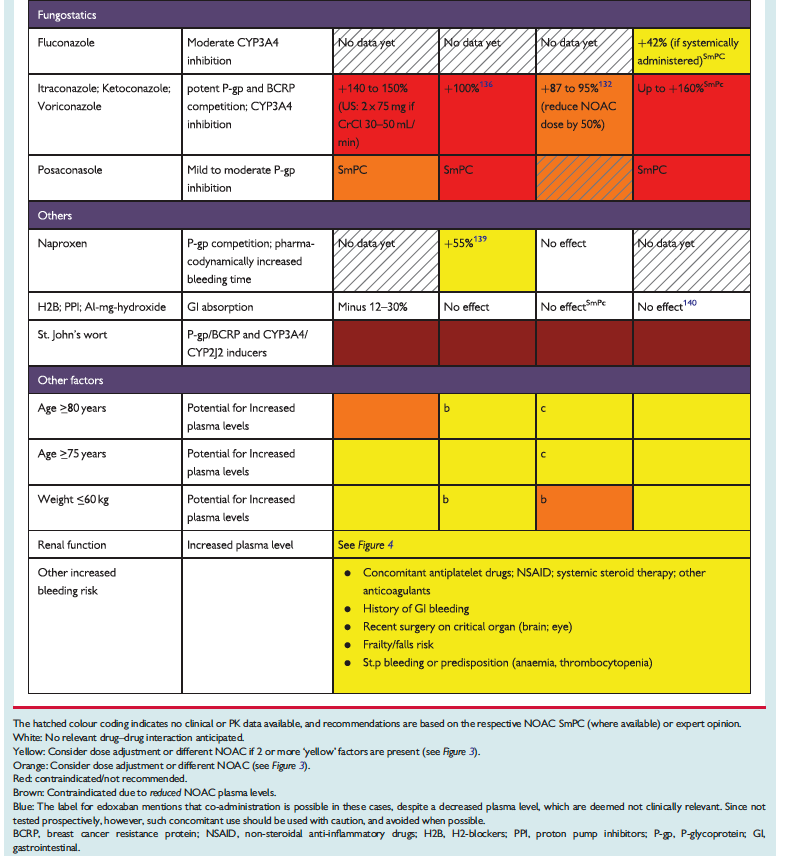
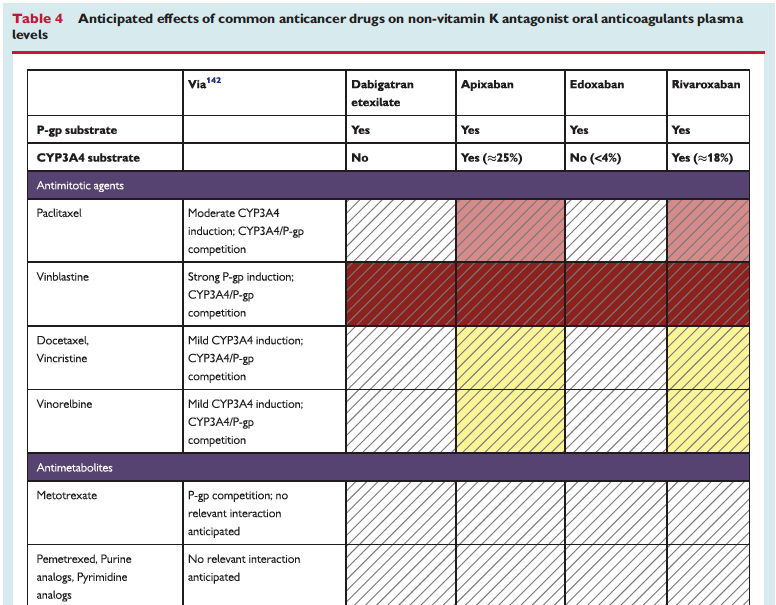
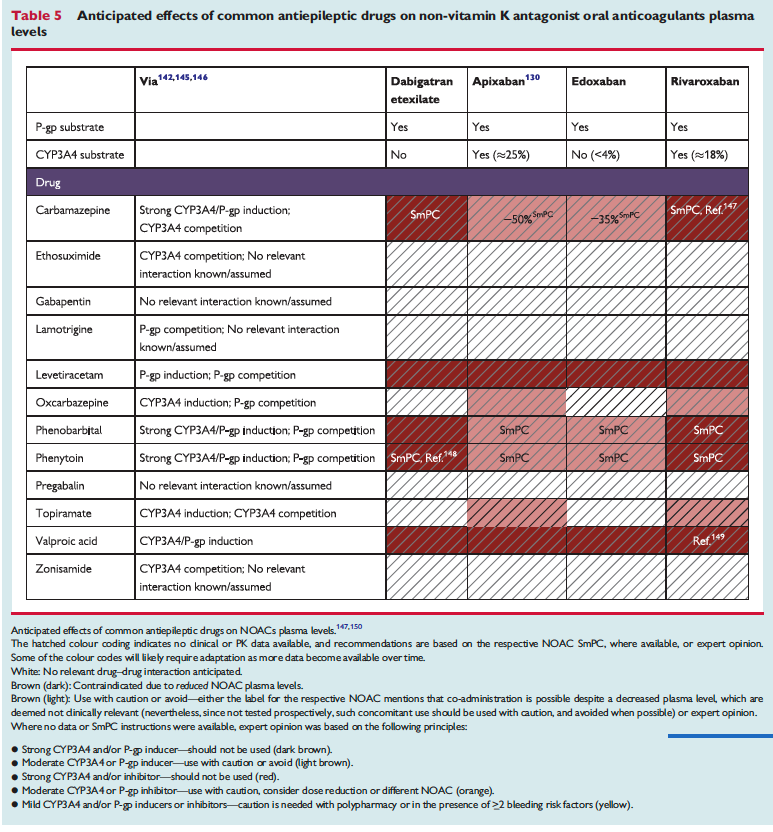
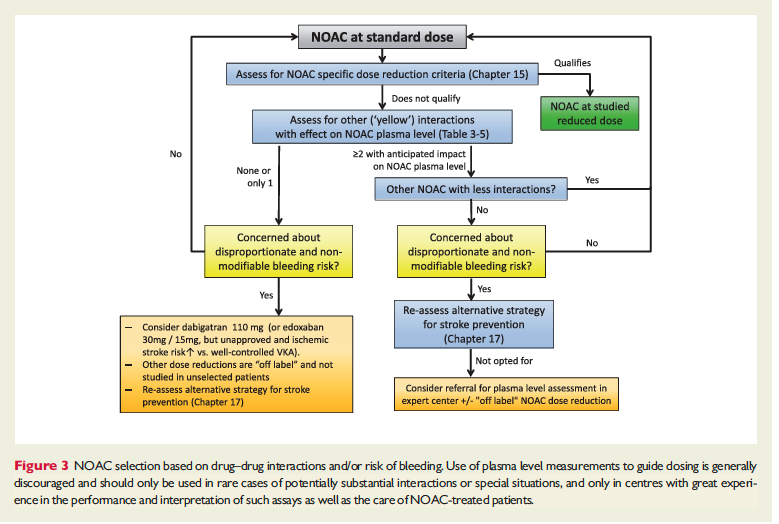
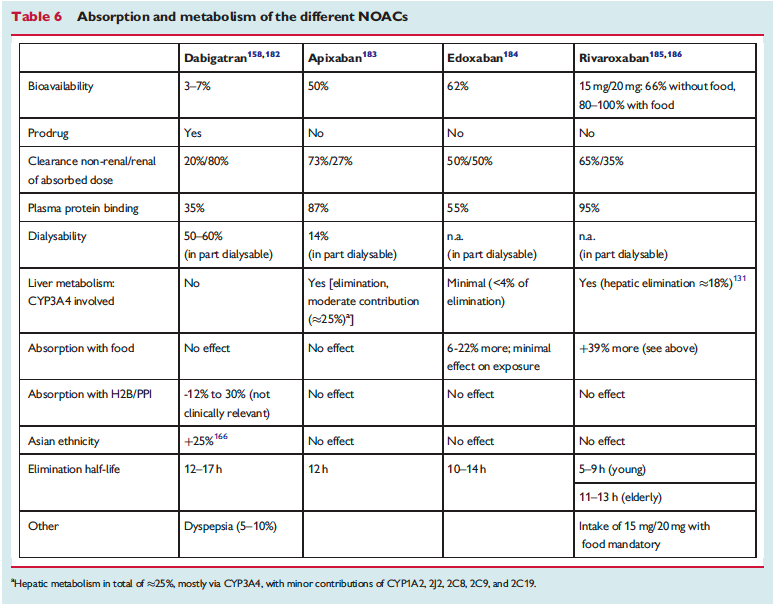
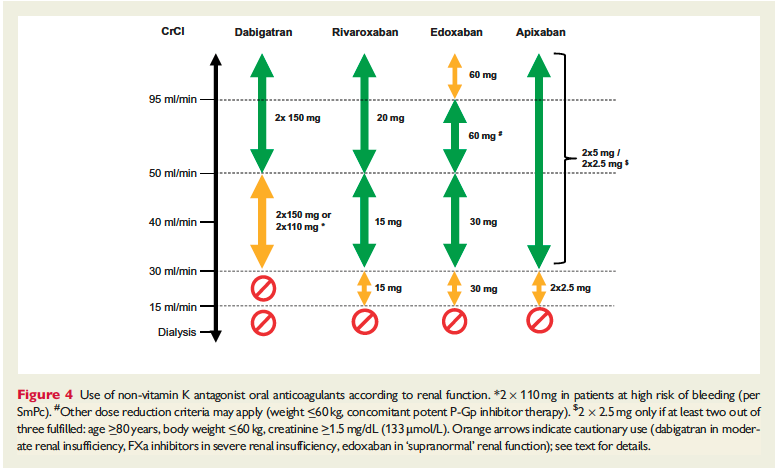
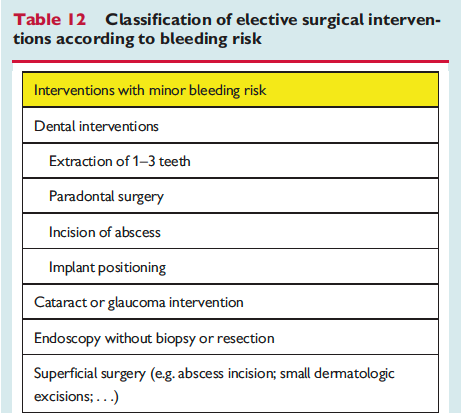
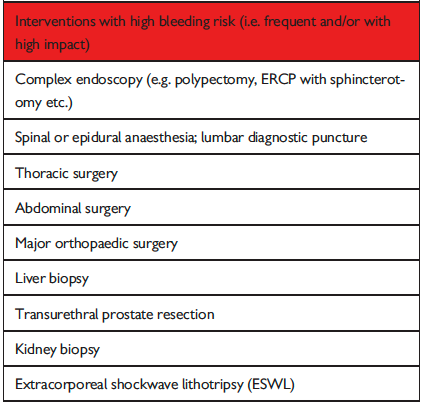
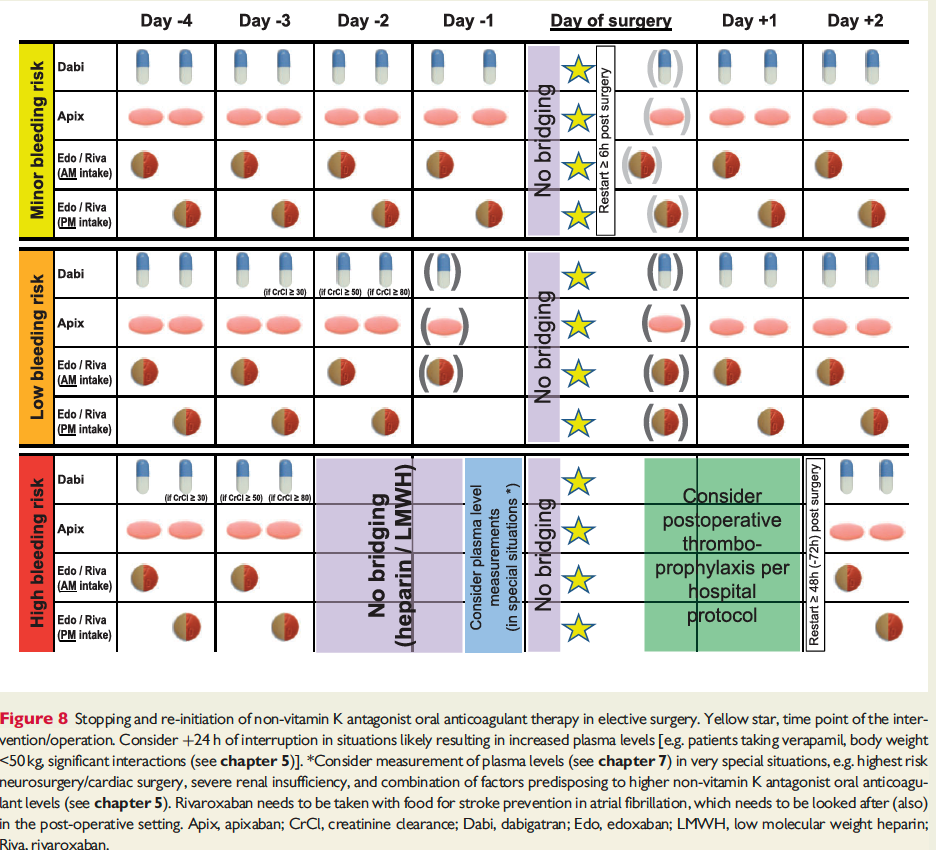
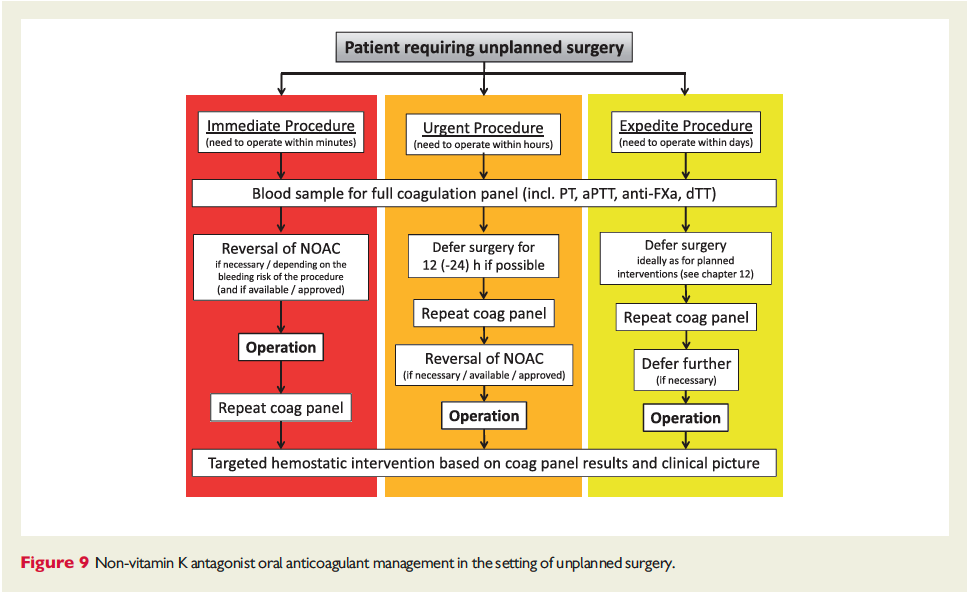
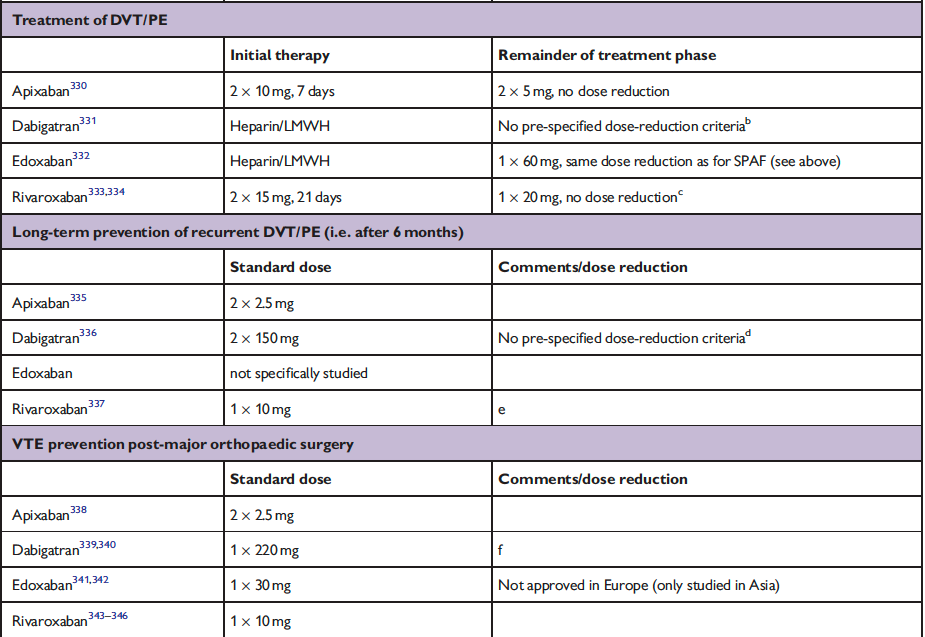
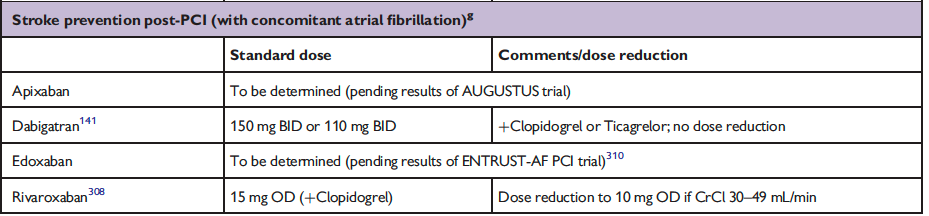
The full set of guidelines from EHRA 2018 are available but the best part of the guidelines are the tables and figures which appear below!
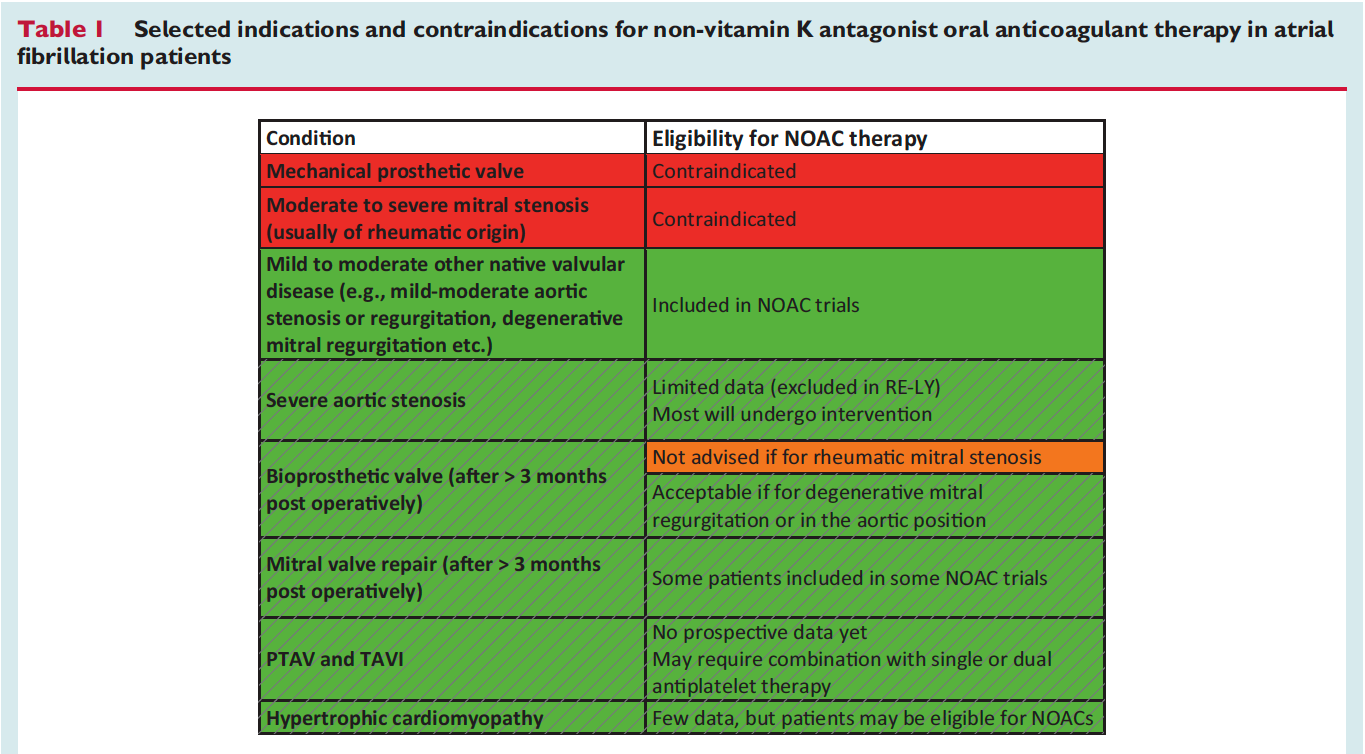
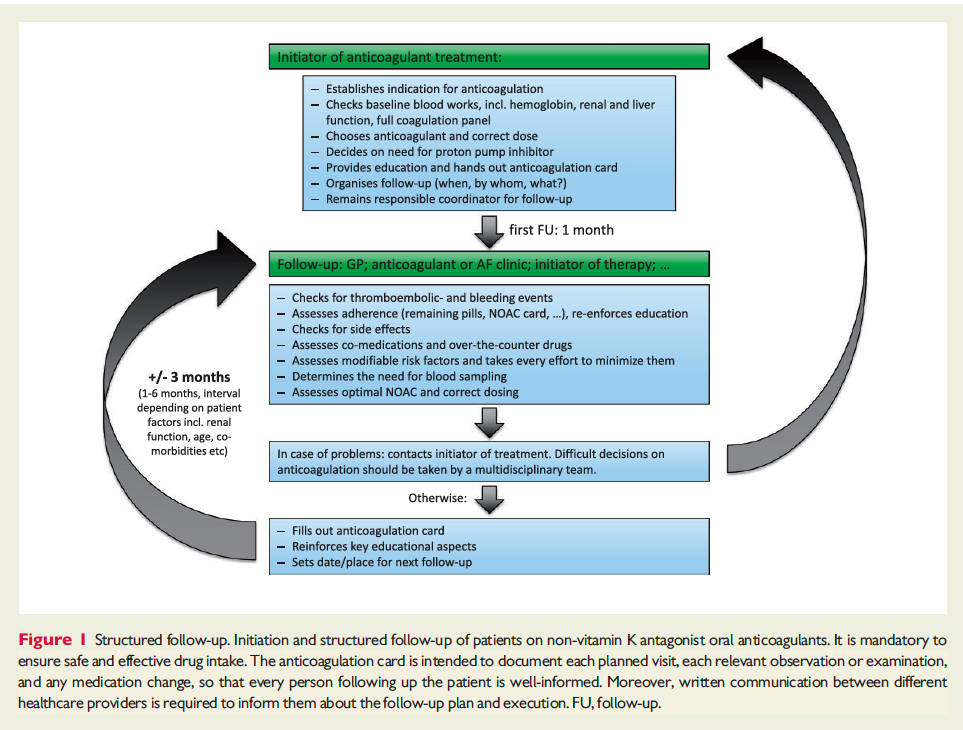
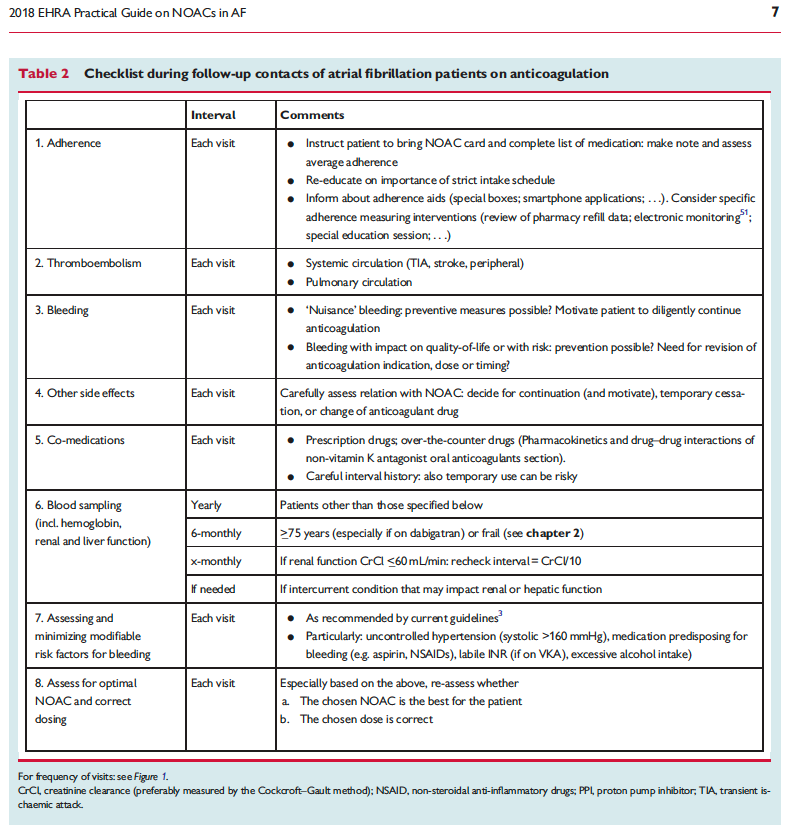
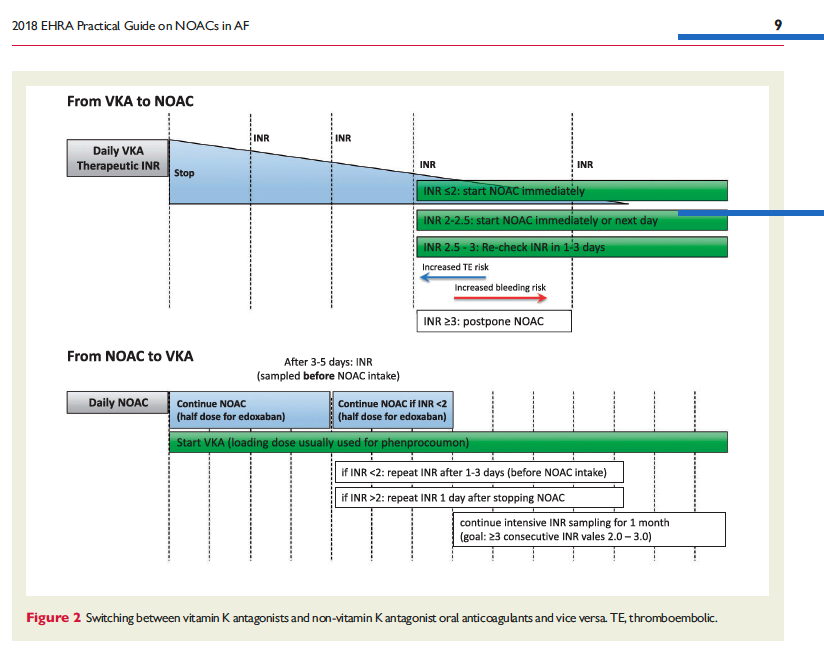
![]()


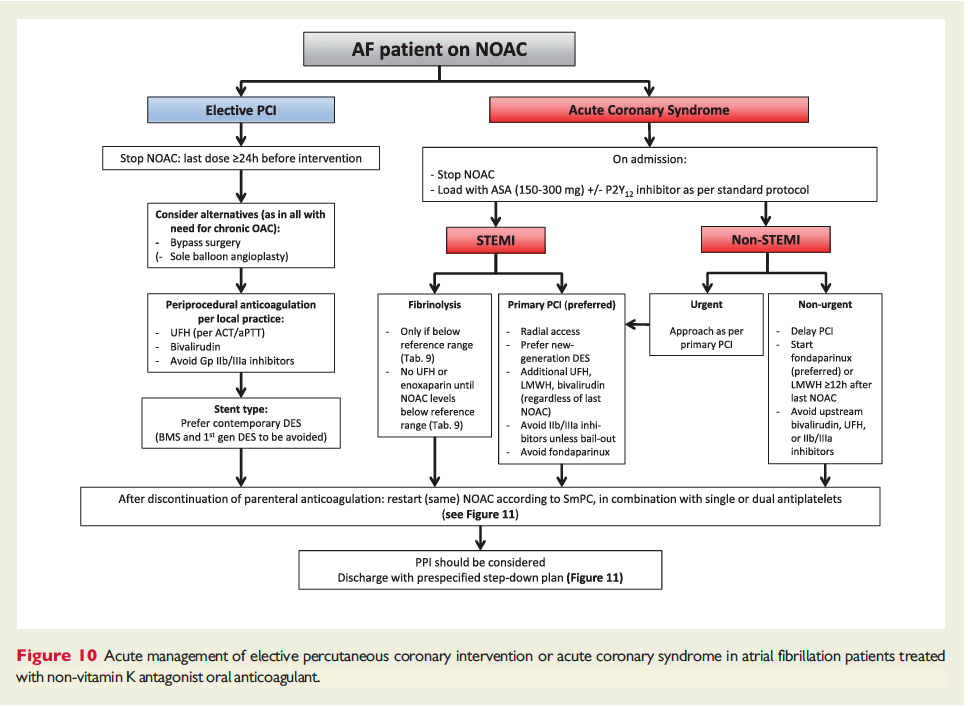
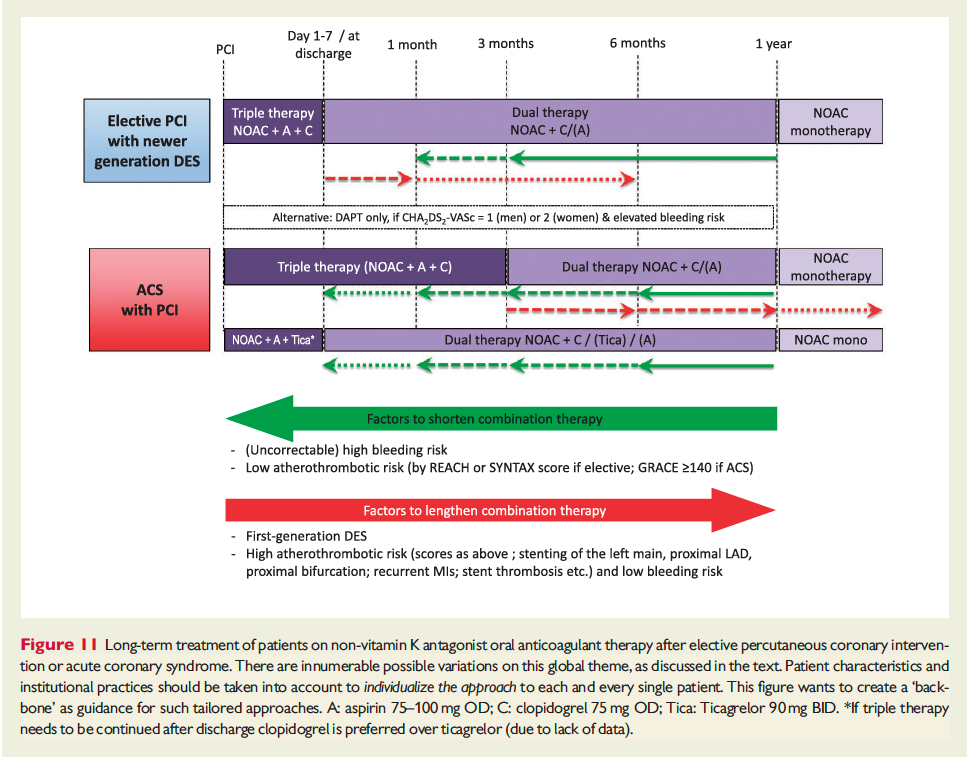
![]()
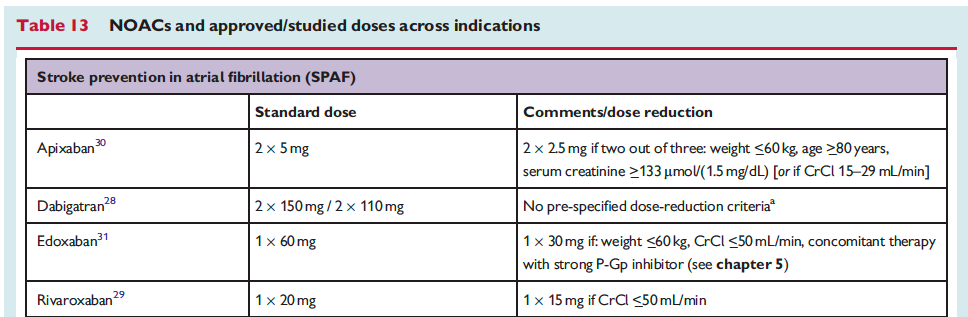
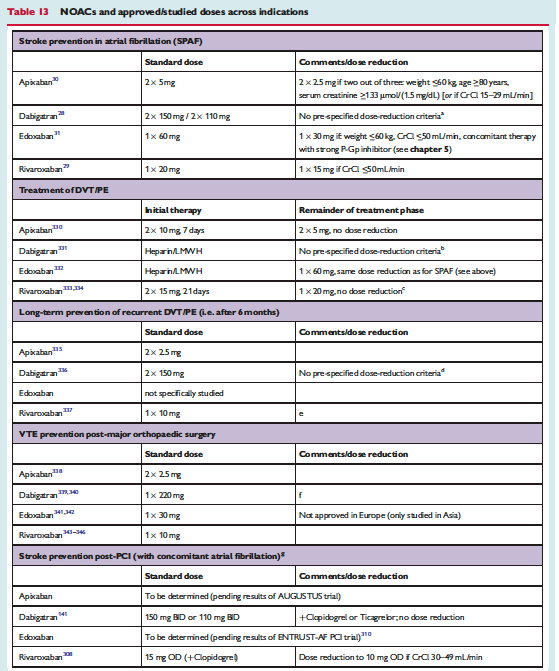
![]()